Written by: Beth Rousseau
Editors: Madeline Barron, Will Dana, and Austin Shannon
New and flashy vaccines tend to get all the press, whereas less attention is paid to safe, effective, and well-established vaccine technology, such as vaccines derived from virus-like particles (VLPs). VLP-based vaccines have been on the commercial market for years and include those used to prevent diseases like hepatitis B and cervical cancer. The success of these vaccines is rooted in the science underlying their development and activity in the body. With many new VLP vaccines currently in clinical trials around the world, it is evident that, even amidst the wave of new vaccine technologies, VLPs are here to stay.
What are virus-like particles?
VLPs are comprised of the structural proteins that form one copy of a virus, also called “the virion”.

The difference between VLPs and virions is that VLPs lack the viral genome encoding the proteins needed to make more viral particles (Fig. 1). Because of this, VLPs cannot produce more of themselves. As a result, VLPs are safer to handle than replication-competent viruses. Moreover, the proteins that form VLPs can self-assemble into their namesake, a virus-LIKE particle. The property of self-assembly is key to the production of VLPs in bacteria or yeast. Manufacturing VLPs with microorganisms is more affordable than if human cells were required. Low cost and ease of manufacture can make it possible to test many different VLP vaccine formulas in the early stages of vaccine development.
How are VLPs used as vaccines?
When the immune system encounters a foreign molecule (i.e. an antigen), it makes antibodies that bind to the antigen and mark it as an invader so immune cells will destroy it. A viral protein is an example of an antigen. Vaccines that contain viral proteins train the immune system to make antibodies capable of quickly stopping future viral infections. Effective vaccines tend to provoke a strong immune reaction. Because VLPs are composed of many repeats of a viral protein, and therefore contain many copies of the same antigen, they can trigger a robust immune response. This makes them ideal vaccine candidates. Indeed, VLPs have been used as vaccines for decades and have a solid track record of safety and efficacy.
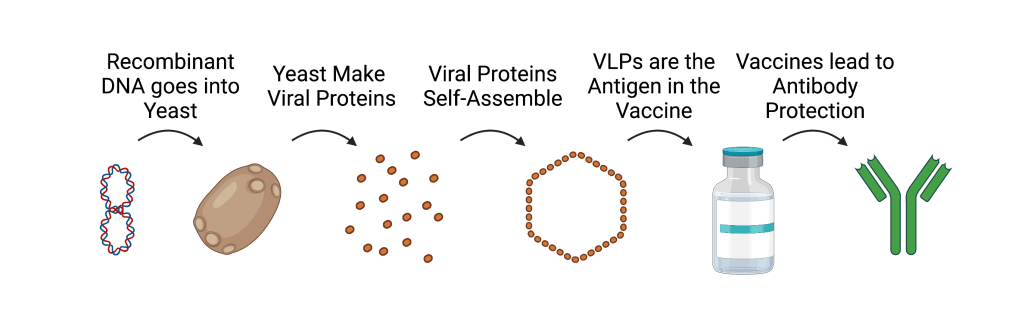
The first commercially available VLP-based vaccine was approved in 1986 to prevent hepatitis B. The vaccine is produced by delivering DNA encoding the hepatitis B surface antigen (HBsAg) gene into yeast (Fig. 2). Yeast use the HepB DNA to create HBsAg proteins. These proteins self-assemble into VLPs approximately the size of the hepatitis B virion, which are then incorporated into vaccines that train the immune system to make antibodies against HBsAg.
The purification of HBsAg VLPs from yeast was a big advancement. The original hepatitis B virus (HBV) vaccines were derived from human blood products which needed careful sterilization to free them from contaminating pathogens. Yeast do not present the same risk of contamination. Furthermore, as mentioned above, viral proteins produced by microbes like yeast can self-assemble into uniform, spherical VLPs. As such, with their homogeneity in form and density of the repeated antigen, the new VLP-based HBV vaccines were purported to better elicit antibodies than their blood-derived counterparts.
Aside from the HBV vaccine, another commercially available VLP vaccine is Gardasil, which protects against human papilloma virus (HPV), the leading cause of cervical cancer. The outermost layer of the HPV virion is formed from two proteins, L1 and L2. Gardasil is made from the L1 proteins of HPV-16 and HPV-18, the two HPV strains that are responsible for a majority of HPV-induced cancers. Because the structure of the L2 protein is less variable between different strains of HPV compared to L1, newer HPV vaccines are being developed that contain both L1 and L2 proteins with the goal of promoting immunity against many HPV strains.
To this point, both the HBV and HPV vaccines are examples of VLPs that contain many repeats of a single viral protein. However, it is possible for VLPs to contain the full complement of structural viral proteins that comprise a virion. These may offer better immunity because antibodies could be made against all of the proteins in a virion rather than just one. As a result, the immune response may be better able to handle viruses that contain mutations or differences from the strain that the person was initially vaccinated against.
What are some of the advantages of VLPs as vaccines versus other types of vaccines?
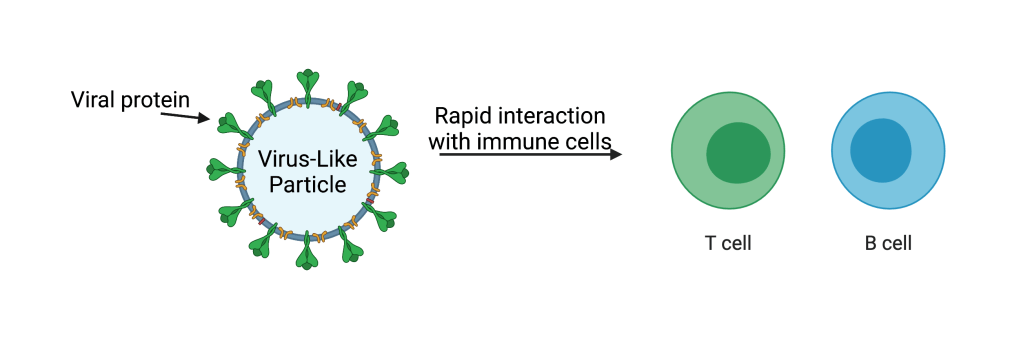
As described, the purpose of a vaccine is to train the immune system to recognize antigens. This must be done safely and effectively. In both aspects, VLPs have certain advantages over other viral vaccine types including attenuated, peptide, mRNA, and viral vector vaccines. Some of the oldest vaccines, such as for yellow fever or polio, are attenuated or inactive viruses, which means they are almost identical to the original virus. Although rare, it is possible for these to revert back to an infectious form. VLPs are safer than attenuated vaccines in that it is impossible for VLPs to convert to an infectious virus. With respect to efficacy, VLPs can outperform peptide vaccines (which contain viral protein antigens that do not self-assemble into structures larger than themselves) in that VLPs tend to elicit greater antibody production from immune cells. While the newer vaccine platforms such as mRNA and viral vectors can also be effective, one potential advantage of VLP vaccines is that the antigen is immediately ready to interact with immune cells at the time of administration. Viral vector and mRNA vaccines necessarily have some delay between the time of administration and the availability of the antigen. Therefore, VLP vaccines induce a robust, more rapid, immune response relative to these other technologies.
What VLP based vaccines are on the horizon?
There are several VLP vaccines in clinical trials around the world, including those for respiratory syncytial virus, Ebola virus, and human immunodeficiency virus. One particularly exciting vaccine candidate against COVID-19 from Oravax is in clinical trials in Israel. In contrast to mRNA-based COVID-19 vaccines, this vaccine does not need to be injected, but, because it is made of protein, can be administered orally and does not need to be kept frozen. Due to the instability of RNA, the currently available COVID-19 vaccines are unlikely candidates for oral delivery or storage at ambient temperatures. The availability of an orally administered COVID-19 vaccine (or any vaccine) would appeal to the many people who are averse to needles while eliminating biologically hazardous needle waste that requires special disposal. Moreover, it circumvents the need for trained personnel to administer intramuscular injections. All of these factors could make it easier to protect people around the world faster and more affordably than current vaccine formulations allow.
Ultimately, VLP vaccines are an established technology with inherent characteristics that make them safer and more effective than some other types of vaccines. For instance, VLP vaccines cannot revert to an infectious form; they present antigens in dense repeats that mimic the native virion, and thus may be more likely to elicit antibodies than lone antigens; and they are immediately available to interact with immune cells upon vaccine administration, eliminating the delay to antigen presentation. Finally, VLPs could be made to incorporate multiple antigens in order to elicit antibodies against even more viral proteins, potentially providing more robust protection. These characteristics make VLPs a trustworthy staple in the exciting world of vaccine technology.
Note: All scientific illustrations used in this article were created using BioRender.com

Beth Rousseau is a PhD candidate in the Department of Biological Chemistry. Her research in the Turner lab centers on engineering virus-like particles for the delivery of CRISPR-Cas9 and prime editing enzymes into neurons and other difficult to transfect cell types. She hopes to find a post-doctoral position in a lab where she can continue to break new ground in genome editing or virology. In her spare time, she likes to do home improvement projects and to cuddle with her two orange cats.
