Author: Kristina Lenn
Editors: Christina Vallianatos, Andrew McAllister, and Sarah Kearns
Spoiler: For a better reading experience, make sure to see the amazing Doctor Strange!
I love Marvel movies, and they’re even more fun to watch as a scientist. In many of them, some energy source is at risk of falling into the wrong hands. Lest the source destroy not only the planet but also the entire galaxy, a bunch of unlikely misfits band together to ensure the energy source’s safety and security. The power source in question is something that has the paradoxical capability of both sustaining and destroying life, like the sun. Extraterrestrial battles take place with the brutish Hulk and the witty turbo-powered Iron Man. (Or, if you prefer Guardians of the Galaxy, you can fight with the smart-aleck Rocket and the cute-yet-somewhat-airheaded Groot.) The bad guys want to use this energy to have unlimited power; the good guys want to harness the energy in a more controlled manner.
Here on the real Earth, we may not be fighting extraterrestrials, but we are trying to develop new and efficient methods for harnessing powerful energy sources to create a cleaner environment. Wind, water, and geothermal energy have proven to be useful and their forms are part of our daily lives. We feel the wind, wash with water, and walk on the Earth’s crust. However, each of these must still be handled with care while trying to create enough energy to power homes and businesses. While these are all natural sources of energy, they are also the sources of natural disasters. Heavy wind and floods are astronomically destructive (Hurricane Katrina, Hurricane Sandy) and hydrothermal power plants increase the risk of earthquakes.
The most powerful energy source we have is the sun, but the main difference between it and the aforementioned methods is that it cannot be directly handled. Remember the Tesseract in Captain America (and later on in the Avengers)? Or, if you’re more of a Dr. Strange fan like me, the Eye of Agomotto? These are energy sources that are so powerful that if their masters do not use them properly, they can be devastatingly destructive. However, if harnessed, they can be formidable, albeit enigmatic, resources. In our world, these allies would be called solar cells.
Solar cells are devices that convert energy from the sun into electricity (the flow of negatively-charged particles known as electrons). There are three types of materials that can be involved in the flow (or lack of flow) of electricity: conductors, semiconductors, and insulators. The difference among these three depends on what is known as a band gap, or the amount of energy required to displace electrons from the valence band to the conduction band in the material (Figure 1). The larger the band gap, the less likely the material will produce a current.
All atoms have a set of orbitals or bands, which describe where electrons are around the atom. The valence bands are where the electrons reside when they are at rest. When the electrons absorb energy, they move into the conduction band. If there is a distance between the two (i.e., no overlap), then a certain amount of energy, which depends on how large the gap is, needs to be provided to cause electrons to jump from the valence to the conduction band.

Conductors don’t have band gaps, which means that current flows readily through them because electrons can easily leave the valence band. Metals like copper, gold, silver, and tungsten are common conductors. Going back to Dr. Strange, remember how the Ancient One used her sling ring so effectively? She was so in tune with it, she barely even needed to think in order to create mystical portals. That’s how a conductor works: little effort is required to create a current.
Insulators (like rubber and wool) have such large band gaps that hardly any current can pass through them. In keeping with the Dr. Strange theme, an insulator would be comparable to Jonathan Pangborn. Like Dr. Strange, he was a patient that suffered severe injuries; while Dr. Strange’s were to his hands, Pangborn had a spinal injury that left him paraplegic… until he went to learn from the Ancient One. However, once he learned how to walk again, he only used his powers for himself and to heal his own body instead of going deeper and learning how to channel his energy to help others. An insulator, like Jonathan Pangborn, stubbornly prevents any current from flowing.
Dr. Strange, however, is in between the two. At the beginning of his training in Kamar-Taj, no matter how hard he tries, he can’t seem to open a portal to save his life. And when he does, he is still only concerned with his own ambitions. However, he does learn to let go of all the thoughts crowding his mind and focus on the task at hand. Slowly, he starts building his power and realizes what he can do to protect the sanctums. That’s what we would call a semiconductor: something that has a band gap but can still be used to conduct a current. And these semiconductors are ideal materials for solar cells because they have a boundary that channels the current in a specific direction.
Silicon is the most popular material for semiconductors because it is so readily available; in fact, sand is mostly silicon dioxide. It also has an ideal band gap to transport charge efficiently and has been determined by theoretical calculations to be one of the most efficient semiconducting materials. However, silicon solar cells have to be manufactured on glass, which is a very delicate and expensive procedure.
One alternative to silicon is organic materials, materials which are mostly composed of carbon and hydrogen. These hydrocarbon-based (hydrogen and carbon) materials are up-and-coming in renewable energy. Instead of being processed onto glass, they can be manufactured onto plastic using roll-to-roll processing, which is a much cheaper method than using glass and silicon.
However, silicon has been able to achieve an efficiency for converting sunlight to electricity of 25%; organic materials have only been able to achieve 10%. That is a significant disparity for how easy it is for electrons to move around. To make it easier, a method was developed called solution shearing, which decreases the distance between the molecules. When the molecules of some of these solar cell materials are closer together, then the orbitals where the electrons reside will have a better overlap. This makes it easier for electrons to move from one molecule to another, thus creating a better current.
Specifically, one of the molecules studied via this method is 6,13-bis(triisopropyl-silylethynyl) pentacene, which is lovingly called 6,13-TIPS pentacene (Figure 2).
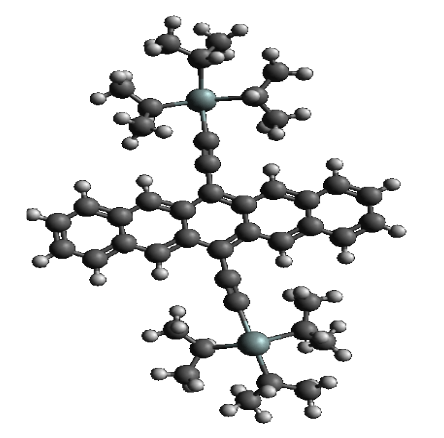
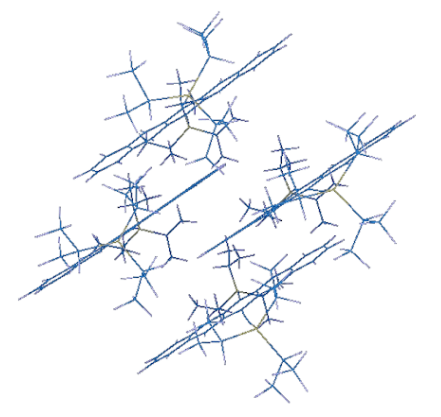
To study how solution shearing can affect the molecule’s ability to transport charge, a unit cell, the most basic structure in a crystal (Figure 3) can be modeled to determine what its best shape will be. Let’s go back to the Tesseract for a minute. It’s also referred to as “The Cube,” and rightfully so because a cube is a type of unit cell. However, not all molecules fit together like a cube where the distance between each molecule is the exact same. Some of the molecules are close together on one side and farther apart on another. Some cause the other molecules to bend and twist while others may not affect the shape of their neighbors at all.
Why does the shape of the unit cell matter? Because that is how we can see where the best overlap occurs between molecules and what the best configuration of the unit cell is for charge transport. The best shape and processing conditions can be determined by using the computer to model different scenarios.
Amazing, isn’t it? The sun is so powerful and massive and can be harnessed to be one of man’s greatest allies. Attempting to do so in a way that is both efficient and safe has proven to be a difficult but worthy task as this will decrease pollution and increase the health of the environment. But what are really in control of the situation are the minutiae of the molecules: how they arrange themselves and interact with each other and adopt different shapes. The power of the universe resides with its smallest particles.
Isn’t it MARVELous?
 Kristina Lenn is a Physical Chemistry and Scientific Computing Ph.D. student in the Geva Lab where she is analyzing the quantum dynamics behind charge transport in photosynthetic reaction centers. She received her B.S. in chemical engineering from Wayne State University and her M.S. in chemical engineering from Cornell University. She spent three years as a lecturer at Wayne State before starting her Ph.D. at Michigan. When she is not busy doing research or writing her next post for MiSciWriters, she is working with the Museum of Natural History as a Science Communications Fellow, volunteering for STEM outreach events, reading as many books as possible, playing friendly games of chess, and writing for her own blog, Chic Geek and Chemistry Freak.
Kristina Lenn is a Physical Chemistry and Scientific Computing Ph.D. student in the Geva Lab where she is analyzing the quantum dynamics behind charge transport in photosynthetic reaction centers. She received her B.S. in chemical engineering from Wayne State University and her M.S. in chemical engineering from Cornell University. She spent three years as a lecturer at Wayne State before starting her Ph.D. at Michigan. When she is not busy doing research or writing her next post for MiSciWriters, she is working with the Museum of Natural History as a Science Communications Fellow, volunteering for STEM outreach events, reading as many books as possible, playing friendly games of chess, and writing for her own blog, Chic Geek and Chemistry Freak.
Read all posts by Kristina here.
