Written By: Christine Lu
Edited By: Christina Del Greco, Peijin Han, and Emily Glass
When you think about cancer, what pops into mind? Likely pain, disease, death, or even a family member who has been affected by cancer. In terms of the cause of cancer, we most likely think of genetic mutations. Very few of us will think about epigenetics. Yet, this does not diminish the fact that epigenetics takes on an equally important role in cancer progression. To understand the role of epigenetics in cancer, we must first appreciate what epigenetics is.
Here is an interesting question that will introduce us to epigenetics: Why is a cell from our eyes so different from a cell from our toes even though they both originate from a stem cell? (Figure 1)
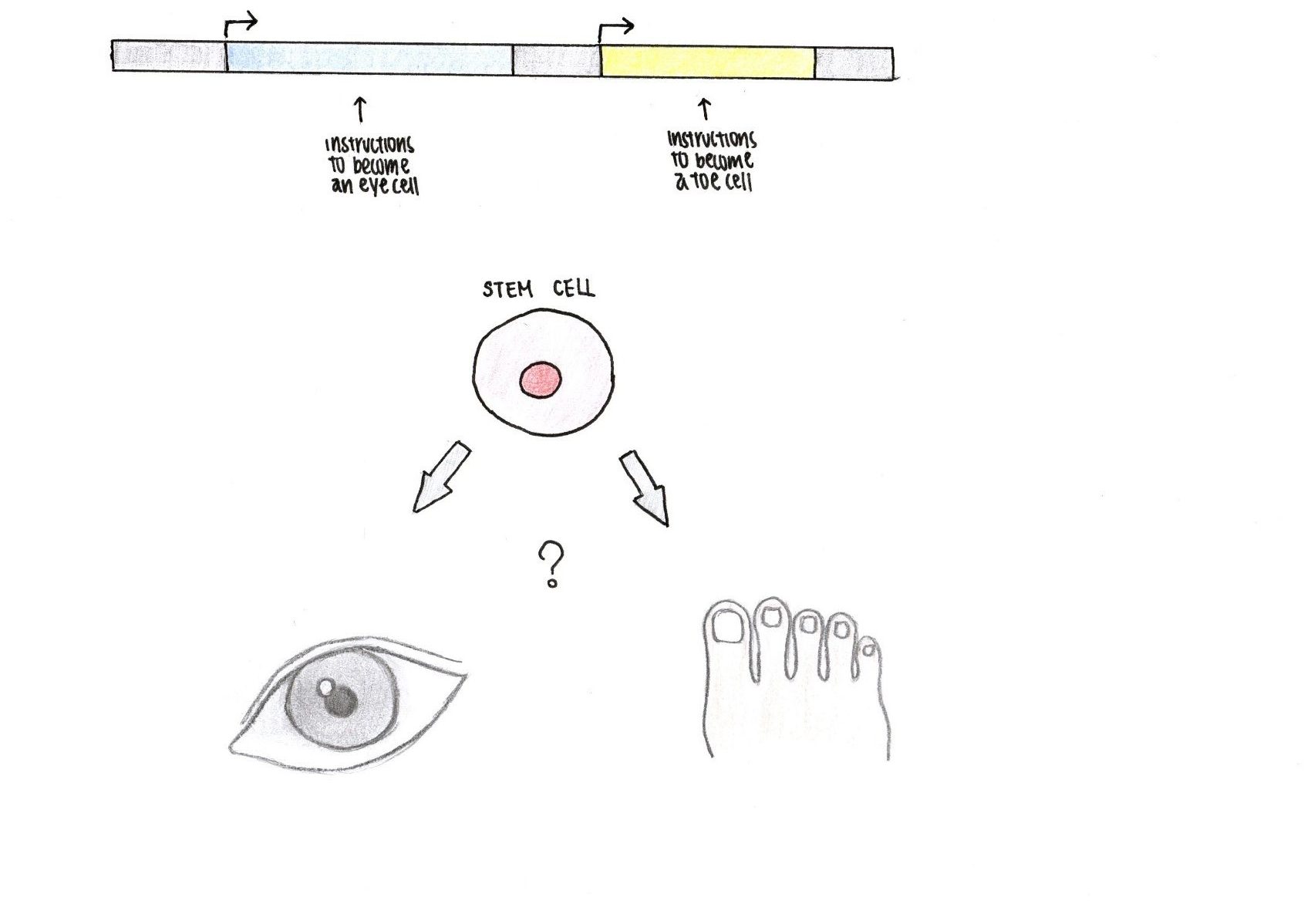
The most straightforward explanation for this is because we all started as one single cell at conception that divided into trillions of cells across development. We know that most of our cells contain the same DNA. If most of our cells, including the cells in our eyes and our toes, contain the same DNA, then why are these two types of cells so drastically different from each other?
While the DNA contained in our cells is the same, gene expression varies depending on the structure and function of the cell. We can think of the DNA as a library. While our cells all have the same library, different types of cells use distinct books from the library to obtain the instructions necessary to construct and maintain the cell so that it can perform its designated function.
The human DNA is a huge library containing thousands of books; how do our cells know which books to read? This is where epigenetics comes into play. Let’s break apart the word to better understand its meaning. The Greek prefix epi- means above. The word epigenetics taken together means above the DNA sequence. To simply put it, epigenetics regulates gene expression without altering the DNA sequence. How does it work? To explain, let us think of epigenetics as the librarian in charge of the cell’s library. For example, the librarian can help pick and open certain books to guide the cells. At the same time, the librarian can also censor some books from the library to limit the cells from accessing other instructions. We call these epigenetic modifications. Epigenetic modification is defined as “heritable changes made to the DNA, which regulate how genes are expressed, but do not affect the nucleotide sequence itself.”
I will focus on two types of epigenetic modifications in this blog post: DNA methylation and histone acetylation.
DNA Methylation and Histone Acetylation
To simplify things a bit, we will think of our DNA as a short sequence containing only two genes (Figure 2). One gene holds all the instructions that are necessary for the cell to become a cell in the eyes. Another gene contains all the instructions that are necessary to code for a cell in the toes. This means that we have two books in our instruction library, and gene expression produced using either of these books will specialize the cells for their function in each body part.

DNA Methylation
DNA methylation is an epigenetic modification that suppresses gene expression. It is our cells’ way of censoring the books in the library that are unnecessary.
There is a new character in play critical to our understanding of DNA methylation and, really, all epigenetic modifications: the RNA polymerase. You can think of the RNA polymerase as the workers in the cell that read the books from our library. They then pass the information down to other workers in the cell in a process called transcription. The other workers, more precisely the ribosomes, will use the information passed down to make proteins in a process called translation. The proteins in our cell will define the function of the cell. For instance, the proteins in our eye help us process what we see and the proteins in our toe help generate movement. DNA methylation will physically prevent RNA polymerase from transcribing genes that do not aid the cell’s specified function (Figure 3). In the figure, our cell receives chemical signals to become a cell in the eyes. Thus, the gene encoding the information to make an eye cell is not methylated. RNA polymerase can bind to the non-methylated eye gene, allowing eye gene expression. Meanwhile, RNA polymerase cannot bind to the methylated toe gene, preventing toe gene expression. Importantly, notice that DNA methylation occurs on the DNA and does not affect the DNA sequence.

Histone Acetylation
Another type of epigenetic modification, aside from DNA methylation, is histone acetylation (Figure 4). Before we discuss histone acetylation, we need to know how histones work. Our DNA winds around histones to pack itself so that it does not take up too much space in our cells. When DNA is in its compact form, RNA polymerase cannot access the genetic information within, suppressing gene expression. Think of it as a library where all the books are closed and shelved.
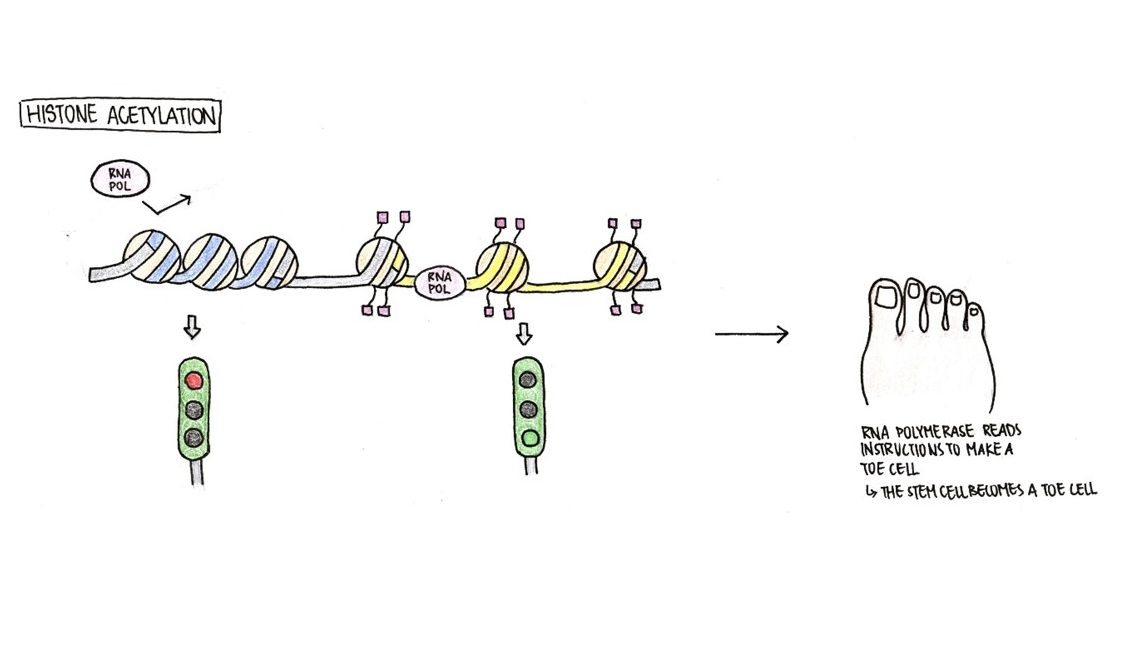
Histone acetylation promotes gene expression. When acetylation markers are placed onto histones, DNA cannot wind itself as tightly around them. Now, the RNA polymerase can access the genes. Continuing with our analogy, we can think of this as a library where the librarian has helped us pick out the books that we need and opened them for us. Under this condition, the RNA polymerase can easily find the correct information required. In Figure 4, our cell received signals to become a toe cell, therefore the histones around the toe gene are acetylated. Because of the acetylation markers, the toe gene around the acetylated histones is uncoiled, allowing RNA polymerase to bind and transcribe the gene.
Epigenetics and Cancer Cells
In the few paragraphs above, I have explained what epigenetics is and how it regulates gene expression in our cells to help them specialize and carry out their respective functions. What is really exciting about this topic is that DNA methylation and histone acetylation can be applied to cancer to explain tumor growth.
Our DNA contains tumor suppressor genes that will induce cell death when an abnormality in the cell is detected. This prevents the cell from massively replicating itself and becoming a tumor. However, epigenetic abnormalities are often detected in cancer cells, including excessive DNA methylation and lack of histone acetylation of the tumor suppressor gene, which prevents gene expression. We can go back and think about this in terms of our analogy. Imagine the tumor suppressor gene as our book in the library. An excess of DNA methylation and an absence of histone acetylation will store away our book to prevent RNA polymerases from accessing the instructions for tumor suppressors (Figure 5). With less tumor suppressor gene expression, the cancer cell escapes cell death.
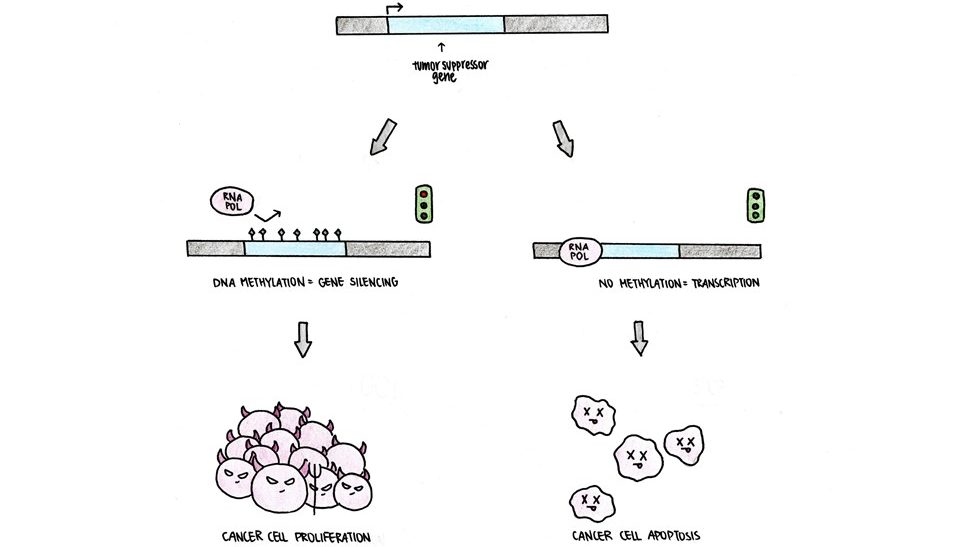
Fortunately, with the understanding of epigenetics and cancer, researchers are moving forward with new treatments to reverse these epigenetic abnormalities found in cancer cells. For example, in the scenario above, developing ways to remove DNA methylation or increase histone acetylation could restore the expression of tumor suppressor genes to limit tumor growth or even remove the tumor entirely.

Christine Lu is currently an undergraduate at the University of Michigan, majoring in Molecular, Cellular, and Developmental Biology. She is interested in studying the impacts of environmental stressors on epigenetics and lifespan. She has also done research investigating Hepatitis C viral replication. Outside of biology, she enjoys learning about how social identities shape individual experiences, especially in the public health system. Some of her hobbies include drawing, baking, and solving puzzles.
