Author: Madeline Barron
Editors: Genesis Rodriguez, Alyse Krausz, and Emily Glass
Bacteria are bubbly organisms—literally. As they go about the business of living, many bacteria pinch off little blebs of their outer membrane to form outer membrane vesicles, or OMVs. OMVs are tiny orbs (about 4,000 times smaller than the width of a human hair) that pack a big functional punch. They contain proteins that scavenge nutrients for bacteria to eat, serve as “decoys” that bind up antibiotics and protect bacteria from certain death, and deliver compounds to host cells that cause disease and trigger an immune response. To this end, scientists have sought to exploit the immune-stimulating power of OMVs to generate vaccines that help protect people from bacterial infections. Thus, OMVs may be small, but they could be a mighty weapon to help us keep bacterial pathogens at bay.
How OMVs are Made
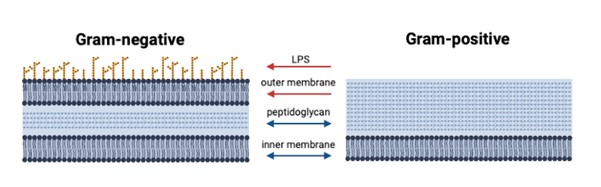
Bacteria are divided into two main classes depending on their structure: Gram-positive and Gram-negative (Figure 1). OMVs are classically associated with the Gram-negative bunch. This is because Gram-positive bacteria do not have an outer membrane—their inner membrane is covered with a thick, protein-rich coat called peptidoglycan. Gram-negative bacteria (E. coli is a well-known example) also have a peptidoglycan layer, though theirs is draped with an additional membrane that interfaces with the environment. Long molecules, called lipopolysaccharides (LPS), decorate the outer membrane surface and are one way in which our immune cells interact with bacteria.
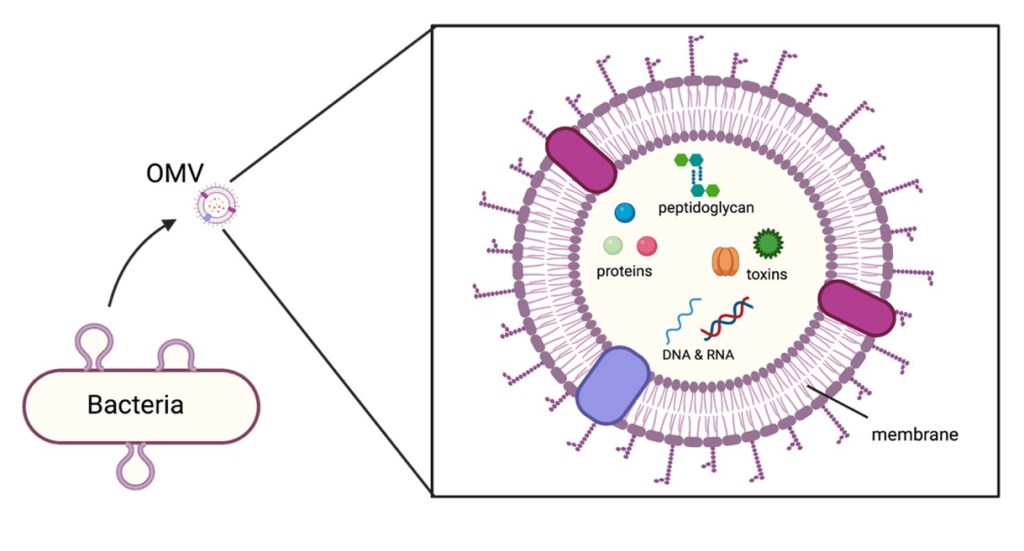
The outer membrane is tethered to peptidoglycan through a number of protein crosslinks, and OMVs are produced when there is a reduction or change in these crosslinks. In such cases, the membrane balloons away from the cell and intracellular components, like cytosolic proteins, peptidoglycan fragments, and DNA, among other molecules, are packaged into the membrane bleb (Figure 2). After it is loaded up, the OMV is pinched off, free to float away into the surrounding environment.
How many OMVs bacteria produce, and what is packaged inside, varies depending on their surroundings. Factors like temperature, nutrient availability, presence of other bacteria, and antibiotics have all been shown to influence the rate of OMV production and cargo composition. The process by which cargo is selected for packaging into OMVs is not well understood. What is known is that OMVs allow bacteria to interact with their environment and send components from inside the cell into the outside world.
OMV Functions
The functions of OMVs are as varied as their composition. For instance, dispose of toxic products accumulating within the bacterial cell, like little cellular garbage trucks. Sometimes instead of acting as waste containers, OMVs help bacteria find food. For example, they can contain proteins that latch onto iron, a critical nutrient for bacterial survival within a host, within the environment so bacteria can easily access it. In addition to assisting their “parent” bacterial cell, OMVs can also disperse within the environment and promote the survival and growth of a broad range of bacteria. Indeed, some bacteria that live in our guts release OMVs harboring enzymes that break down sugars for other intestinal bacteria to feast on. To this end, OMVs are a means by which members of a diverse bacterial community can interact and communicate with one another.
While OMVs are secreted by friendly and disease-causing bacteria alike, they have been primarily studied in the context of the bad guys, and for good reason. OMVs derived from bacterial pathogens contain virulence factors that enhance bacterial survival within and/or damage their host. To the first point, OMVs can serve as decoys that bind up antibiotics or host-derived, microbe-killing compounds to protect the pathogen cell. They can also carry enzymes that tinker with antibiotic molecules to render them inactive—a bacterial bomb squad, if you will.
In contrast to their role in protecting pathogens, OMVs shuttle around compounds, like toxins, that harm host cells. OMVs shelter toxin molecules from host enzymes that might degrade or damage them, so they can safely reach their target cells. For instance, Vibrio cholerae, which causes cholera, a disease characterized by severe diarrhea and dehydration, releases OMVs filled with a toxin that interacts with intestinal cells to induce fluid accumulation in the gut. The deliverance of damaging compounds to host cells by OMVs is characteristic of many other bacterial pathogens, including those that infect body regions beyond the gut.
OMV Vaccines and Meningitis B
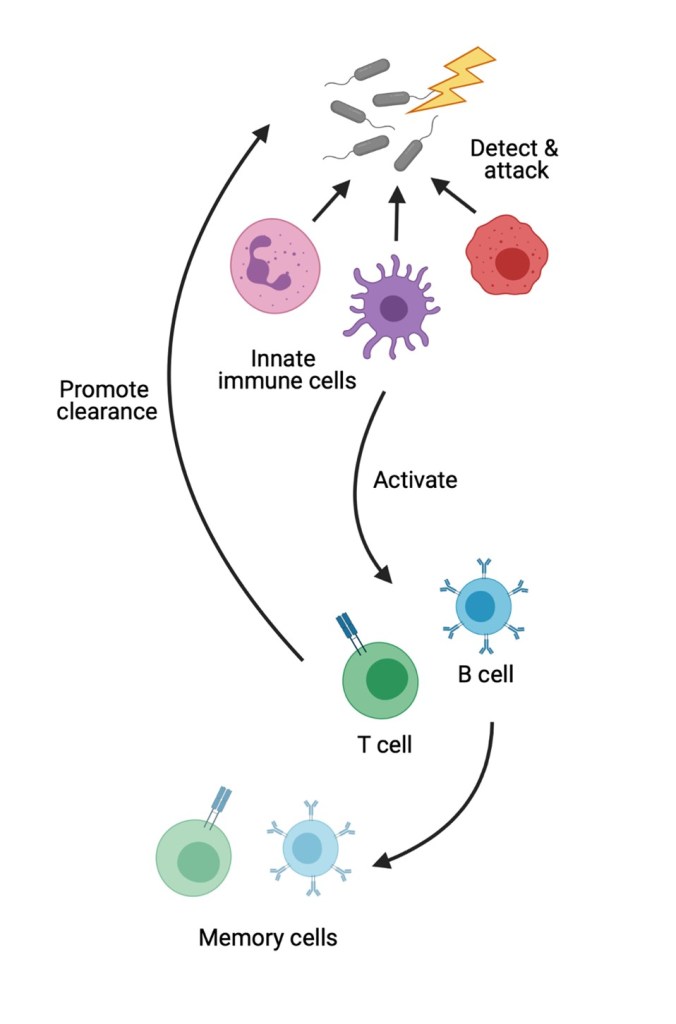
Besides acting as shuttles for virulence factors, OMVs possess components of bacterial cells that can activate and amplify the innate and adaptive immune responses (Figure 3). The innate immune response is our first line of defense against pathogenic invaders. Innate immune cells recognize conserved features of bacteria, such as common molecules on the bacterial cell membrane, like LPS. When they detect a pathogen, innate immune cells battle the invaders while alerting adaptive immune cells (i.e., B and T cells) of the pathogen’s presence. These cells recognize pathogen-specific molecular features and orchestrate a response to eliminate the invader while generating memory cells that will remember that pathogen, and how to deal with it, the next time it comes around. These memory responses are the basis of vaccines, which give the immune system a little taste of a pathogen so it can learn how to fight the real thing.
The immune-stimulating nature of OMVs has led scientists to wonder: Can we purify OMVs and use them to generate vaccines against bacterial pathogens? Indeed, a good vaccine should activate both arms of the immune response without actually causing disease, be stable under diverse conditions (i.e., different temperatures), and contain molecular features of the “real” pathogen for the immune system to recognize. OMVs have all these features and more.
With that, OMV-based vaccines have been investigated for a number of pathogens, though many have yet to make the leap from studies in animal models to humans. There are success stories, however. One example is an OMV-containing vaccine, called Bexsero®, approved for preventing meningitis B. Meningitis is a relatively rare disease characterized by swelling of the brain and spinal cord, as well as severe bloodstream infections. The disease is caused by the bacterium Neisseria meningitidis, which can live in the nose and throat and spread to others via coughing, sneezing, sharing saliva, or living in close quarters. Individuals can succumb to severe infection quickly, sometimes within 24 hours. Given this short window for treatment, preventing infection via vaccination is key.
There are five main groups of N. meningitidis that cause disease (A, C, Y, W, and B) that differ slightly based on the molecules on their surface. Vaccines against ACYW N. meningitidis have been around for years. They target specific sugars on the outside of the bacterial cell. With group B, this was not an option; the sugars on these bacteria look a lot like molecules found in the human brain. A vaccine targeting the bacterial surface sugars of B would increase the likelihood of immune cells mistakenly recognizing and attacking the look-alike brain molecules. For this reason, scientists looked to OMVs.
Along with other proteins involved in N. meningitidis pathogenesis, Bexsero® contains OMVs adorned with bacterial surface proteins that activate and amplify the immune response against N. meningitidis. And the vaccine works! Multiple studies have demonstrated that it protects against meningitis B, including one conducted in Quebec in which over 48,000 people between the ages of 2 months and 20 years (the most susceptible age range) were vaccinated with Bexsero®. None of the vaccinated individuals developed meningitis B, whereas several cases cropped up in the unvaccinated individuals.
Caveats of OMV-based Vaccines and Moving Forward
For all their potential as vaccine candidates, OMVs do have their pitfalls. For instance, some molecules on the surface of OMVs, namely LPS, can be too immune-stimulating. That is, they induce such a robust response that it becomes highly damaging, and potentially deadly, to the host. The variability of OMV composition can also present a challenge. If a vaccine is developed using OMVs derived from, for instance, strain 1 of a pathogenic bacterium, it does not necessarily mean it will be protective against a closely-related strain 2, which could have slightly different surface or intracellular molecular structures than its vaccine-targeted counterpart. Finally, sometimes OMVs possess molecules that do not activate an immune response but interfere or suppress it. This is the exact opposite of what we want a vaccine to do.
However, all is not lost. Many bacteria are genetically malleable, which means scientists can tweak their DNA to alter and select for the proteins present on and within OMVs. In doing so, they can increase the safety and efficacy of OMV-based vaccine candidates. Moving forward, the ability to genetically engineer OMVs will allow us to better take advantage of all these small membrane blebs have to offer when it comes to vaccine development and infection control.

Madeline (Maddie) is a Microbiology and Immunology PhD candidate in Vince Young’s lab where she studies how the intestinal environment modulates bacterial colonization of the gut. When Maddie is not in the lab, she is writing about science. Given the results of scientific research affect and belong to everybody, Maddie is passionate about making them accessible and digestible to diverse audiences. She has written for Massive Science, the American Society of Microbiology blog, and her personal science blog, Supplemental Material. You can find her on Twitter.
