Written by: Madeline Barron
Editors: Christian Greenhill, Kristen Loesel, and Peijin Han
We are currently at war with antibiotic-resistant bacteria—and it’s not looking good. In 2019, the Center for Disease Control and Prevention (CDC) estimated that 2.8 million antibiotic-resistant infections occur in the United States each year, resulting in 35,000 deaths and billions of dollars in healthcare costs. This is over 28% higher than the approximated number of infections and deaths in 2013. Yet, despite the rise in antibiotic-resistant infections, antibiotics remain our primary weapon for combatting bacterial pathogens; if they stop working, infections that were once easily controlled could become untreatable. Thus, there is a critical need to look beyond our arsenal of antibiotics for new methods to treat bacterial infections.
Enter: Nanoparticles.
Nanoparticles are miniscule structures made up of everything from phospholipids to silver and exhibit promising antibacterial functions. They come in a diverse array of shapes, sizes, and compositions and kill bacteria in various ways, ranging from puncturing cell membranes to inducing production of toxic compounds. Where current antibiotics fall short, such as in treating intracellular bacterial infections, nanoparticles step up to the plate. However, for all their promise, there is still a lot left to learn before nanoparticle antimicrobials are widely adopted in the clinic. Nevertheless, in time, nanoparticles could be the next big thing for mitigating difficult-to-treat bacterial infections.
Antibiotics are becoming less effective at treating bacterial infections
Antibiotics do their dirty work in several ways. They inhibit enzymes that build bacterial cell walls, bind bacterial ribosomes to inhibit protein synthesis, and interfere with DNA synthesis and replication of bacterial cells. A handful of antibiotics are effective against many types of bacteria, while others are narrower in scope. Which antibiotic a doctor prescribes depends on the bacteria causing the infection, as well as other factors like infection severity and potential side effects.
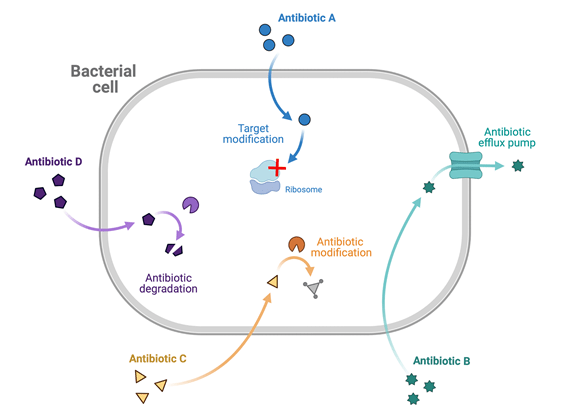
Bacteria are not entirely at the mercy of the drugs we throw at them; they develop strategies for resisting our attacks (Figure 1). For instance, some bacteria secrete enzymes that alter or degrade antibiotics, thus rendering the drug inactive. Others shoot antibiotic molecules out of the cell through efflux pumps embedded in their membrane. Mutations in the cellular targets of antibiotics, such as bacterial ribosomes, can also prevent drugs from binding and killing bacteria. Over time, these mutations will be ‘selected for’ and dominate a bacterial population. People can inadvertently facilitate this process by failing to take their full course of antibiotics, which exposes bacteria to just enough of the drug to select for resistant cells. Overprescribing of antibiotics also promotes the development of antibiotic-resistant bacteria by exposing microbes to the drugs more frequently.
Bacterial resistance to one antibiotic is bad enough—resistance to more than one is downright scary. Indeed, the emergence of multi-drug resistant (MDR) bacteria, such as methicillin resistant Staphylococcus aureus (MRSA), has limited our ability to fight infections caused by these antibiotic-resistant organisms. Moreover, bacteria are notoriously generous with their genes. They can swap pieces of DNA, including those encoding antibiotic resistance, between cells to increase the number and diversity of antibiotic-resistant microbes.
Ultimately, thanks to antibiotic resistance, our trusty chemical weapons of bacterial destruction may not be enough moving forward. We need more options.
The solution to the big antibiotic-resistance problem, however, might be very tiny.
What are nanoparticles and how do they work?
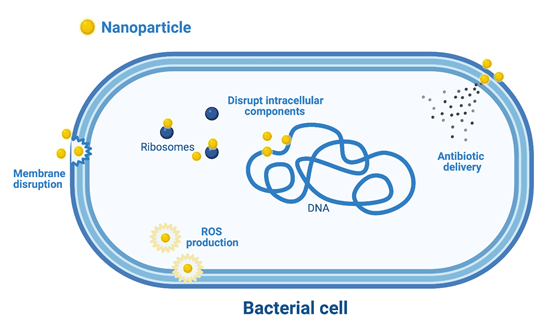
Nanoparticles are, well, nano. Ranging in size from 1-100 nanometers (i.e. 100,000-1,000 times smaller than the thickness of a single sheet of paper), these miniscule particles are made from organic or inorganic materials, like phospholipids or metals, respectively. Nanoparticles come in all sizes, compositions, and shapes, including spheres and rod-shaped structures. They have emerged as attractive tools for treating bacterial infections (particularly those of the antibiotic-resistant variety), thanks in part to their unique physical properties, numerous engineering possibilities, and demonstrated antimicrobial functions. While these functions vary depending on the nanoparticle, there are several general mechanisms by which these deviously small structures take down bacterial pathogens (Figure 2).
- Disrupting the bacterial cell membrane: Like antibiotics, nanoparticles can target the bacterial cell wall. However, rather than interfering with enzymes that build the wall, nanoparticles physically disrupt or destabilize the wall itself. To this end, some nanoparticles carry a positive charge on their surface. These particles are attracted to the slight negative charge of the bacterial cell surface. By binding the membrane, the nanoparticles throw its charge and structure off kilter, ultimately leading to cell death.
- Damaging intracellular structures: Nanoparticles can slip into bacterial cells to damage intracellular machinery, like DNA or proteins. For example, gold nanoparticles were shown to inhibit proliferation of MDR strains of Escherichia coli and Pseudomonas aeruginosa, a respiratory pathogen, by binding DNA and ribosomes.
- Generating reactive oxygen species (ROS): ROS are unstable byproducts of cellular respiration and metabolism. While bacteria have means of dealing with ROS, an over-abundance of these compounds can become toxic to the cell. Nanoparticles, particularly those made from metals, trigger production of ROS to induce everything from cell membrane damage and protein dysfunction to inhibition of DNA replication and transcription.
In addition to battling bacteria head-on, nanoparticles can shuttle antibiotics to sites of infection. That is, instead of replacing antibiotics, nanoparticles act as their personal chauffeurs. This has several advantages over traditional antibiotic administration, particularly in terms of preventing or slowing development of antibiotic-resistant bacteria. For one, when safely tucked inside nanoparticles, antibiotic molecules are protected from degradation or damage within a host, which can increase drug efficacy and lower dosage requirements. Moreover, nanoparticles can be engineered to target tissues of interest, and to release their cargo in response to specific stimuli associated with sites of bacterial infection, such as a decrease in pH. Such targeted release prevents bacteria from being exposed to low levels of antibiotics that could promote selection of resistant cells.
What are the benefits of nanoparticles?
Nanoparticles pack a lot of good into a miniscule package. While antibiotics generally have one cellular target, a single nanoparticle can damage bacterial cells in multiple ways (e.g., disrupting both membrane integrity and DNA.) This multi-targeted approach of nanoparticles lowers the chances of bacteria developing resistance. Moreover, because nanoparticles work differently from antibiotics, they are immune to current bacterial evasion tactics. For instance, bacterial enzymes that degrade a specific type of antibiotic would be ineffective against, say, a silver nanoparticle whose structure and composition is nothing like that of the antibiotic target. Indeed, nanoparticles have been shown to kill MDR pathogens, highlighting their potential efficacy when that of antibiotics is iffy.
Nanoparticles also have the potential to kill hard-to-reach, and thus hard-to-kill, bacteria. For instance, some pathogens do not just interact with host cells—they live inside them. Because most antibiotics cannot pass through the membrane of host cells, killing these intracellular bacteria is a difficult task. Nevertheless, nanoparticles have the potential to overcome this barrier and attack such hidden microbes, either directly or by releasing antibiotics. Additionally, nanoparticles can kill bacteria living in biofilms. Biofilms are multi-cellular communities held together by a sticky matrix that attach to surfaces within and outside the body (think dental plaque and shower scum). Within biofilms, bacteria are protected from external insults, like antibiotics, which have a hard time penetrating the biofilm goop to reach the bacteria inside. Nanoparticles, on the other hand, can pierce the sludge to kill biofilm inhabitants. This is good news for treating infections by MDR bacteria, as dealing with biofilms created by these organisms can be a clinical nightmare.
Why aren’t we using nanoparticles to treat bacterial infections?
Nanoparticles sound like they have a lot of clinical potential, and they do. In fact, nanoparticles, particularly those made of silver, are already used to coat medical devices, like catheters, and in wound dressings to prevent bacterial growth. However, beyond this, there are no commercially available nanoparticle-based antimicrobials approved for clinical use. There are several reasons for why this could be.
For one, the toxicity of nanoparticles, particularly metal-based varieties, is still being investigated. Organic nanoparticles, like those made of phospholipids, can be broken down into biologically inert components. Metals, on the other hand, may be retained in tissues, like the liver, spleen, and lymph nodes for extended periods of time, leading to potential organ dysfunction associated with metal accumulation. The concern for toxicity is linked with the need to determine nanoparticle dosage, as sometimes the amount used in studies to illustrate a point (i.e., “Look! These particles kill bacteria!”) is not physiologically feasible in humans. Moreover, although development of resistance to nanoparticles is less likely, it is not impossible. There have been reports of bacteria developing resistance to silver nanoparticles, one of the most widely used varieties. The ability to fiddle with nanoparticle structure and formulation to increase efficacy could mitigate some of these concerns.
Despite these caveats, nanoparticles as antimicrobial agents are not doomed—we just need to learn more about them. The motivation to do this, however, is bolstered by the benefits of nanoparticles, the flexibility with which we can engineer them, and the threat of antibiotic-resistant pathogens looming large.

Madeline (Maddie) is a Microbiology and Immunology PhD candidate in Vince Young’s lab where she studies how the intestinal environment modulates bacterial colonization of the gut. When Maddie is not in the lab, she is writing about science. Given the results of scientific research affect and belong to everybody, Maddie is passionate about making them accessible and digestible to diverse audiences. She has written for Massive Science, the American Society of Microbiology blog, and her personal science blog, Supplemental Material. You can find her on Twitter.

2 thoughts on “Nanoparticles may be tiny, but they are the next big thing for fighting antibiotic-resistant bacteria”