Coming to you LIVE from the 3rd annual RNA Symposium: Advancing RNA Bioscience into Medicine. Follow us on Twitter or the tag #umichrna!
Live blogger: Sarah Kearns. Editor: Whit Froehlich.
Most of the work done in your cells is done by complex molecules called proteins. Made up of long chains of amino acids, they are required for the structure, function, and regulation of your body’s tissues and organs. The design of these proteins comes from DNA, the genetic code of life. But the pathway from double helix DNA to protein, as it turns out, is a complicated one.
The Central Dogma of molecular biology describes the process as two steps — DNA is transcribed into RNA, and RNA is translated into proteins. While accurate, this model glosses over the regulation done by the various different types of RNA molecules. One such regulator is microRNAs (miRNAs) which are small RNA molecules that regulate gene expression by interacting with messenger RNA (mRNA), the product of transcription.
Particularly in cancers, miRNAs have been found to bind to mRNA before the mRNA has a chance to be translated into key gene regulator proteins. But as miRNAs play a significant role in the progression of cancer, they can also serve as a tool for prognosis. Dr. Eric Fearon, the director of the UM Comprehensive Cancer Center (which recently received an impressive $150M donation), is looking at how miRNA can be used for early detection and potential treatment of cancer, focusing on colorectal cancer.
The Keynote
Diving into the genetics of cancer, Dr. Fearson introduces Apc which is involved in the development of many cancers by activating a kinase signaling pathway. Mutations in Apc leading to a loss of its N-terminus lead to aberrant activation within cancerous cells. Where the cell’s defense system usually catches incorrect protein, mutant proteins within tumorigenesis can evade the immune system, which further aids in resistance to medical cancer therapies. As Dr. Fearon notes, “if you’ve seen one cancer, you probably haven’t seen one cancer,” because there are so many aberrant mutations that lead to subtle differences in genetics even within one tumor.
Within colorectal cancers (CRCs) in particular, there are specific recurrent somatically mutated genes that typically occur within nucleotide repeats, a group to which the Apc gene belongs. Unlike other cancers, there are many mutations that occur throughout the genome that further drive the gain-of-function of the Ras pathway. These collectively lead to the “big bang” model of tumor growth.
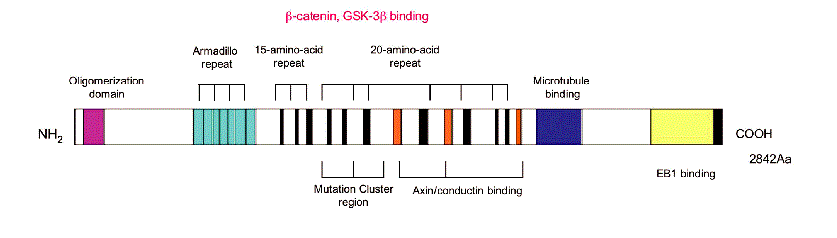
The Apc protein, responsible for tumor suppression, huge for a protein — just under 300 kD. Towards the end of the Apc gene reading frame, the portion of the gene that codes for the protein, there’s a cluster of methionine amino acid (Met) codons towards the C-terminus of the protein. Met also functions as the “start” codon for protein translation so the cluster of them can “confuse” translation to have multiple truncations. The loss of the tail of the protein is a problem because it includes key domains for its function and regulation.
Within the early development of colon cancers, the normal epithelial cells become crypts. Targeting the Apc genes within this cell-type population results in promotion of cell growth and proliferation. But what mechanisms control this?
Looking at a particular signaling pathway (Wnt), β-catenin is an important component of gene regulation of oncogenic transcripts. In the presence of the Wnt signaling molecule, β-catenin gets into the nucleus where it can act as a transcription factor. Without Wnt, Apc prevents β-catenin from becoming a transcription factor by recruiting a proteasome, a complex that breaks down the protein. This is important because Dr. Fearson shows that the knockdown of β-catenin prevents tumorigenesis in mice colons. By turning off and on patterns of expression in Apc-mutant crypts, he further demonstrates the destruction in crypt formation due to proper tumor suppressor function.
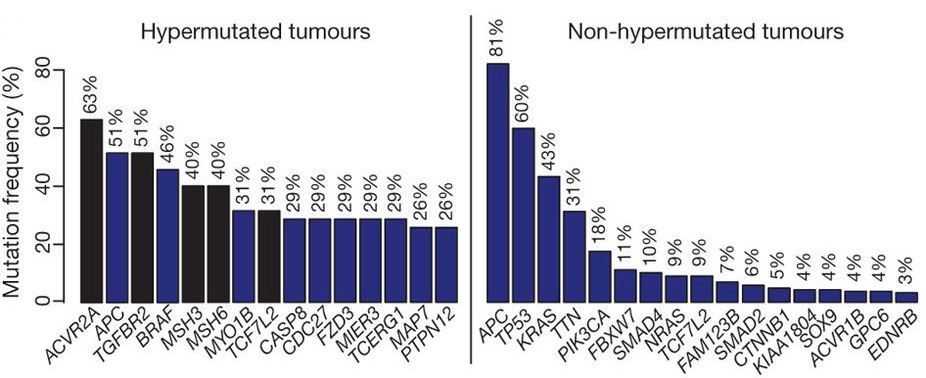
Just like other cancers, colorectal cancers are impacted by alterations in p53 tumor suppressor protein signaling. Dominant negative mutation of the p53 gene is the component that leads to tumorigenesis, and not just the addition of Apc and KRAS. Using CRISPR-Cas9, Dr. Fearon wants to make an accurate mouse model that incorporates all of the mutations that are known to be important in colorectal cancers. By making the transgenic mice with this gene editing tool, his research team can tease out the different interactions of independent proteins.
He concludes his very protein-signaling-based keynote with the hope that better understanding of RNA will aid in discovering the interactions and pathology of the complex progression of cancers. Alluding to his RNA tool development, he suggests that they could also be used for early cancer prognosis.
