By Ada Hagan
Every predator is prey to something. The antelope falls to the lion, the lion falls to the human, and the human, to viruses and bacteria. Bacterial infection is one of the things we fear most. Infections from antibiotic-resistant bacteria can conquer the strongest and smartest of us. But… do the bacteria that live in and around us, that even prey on us, have a predator themselves?
Yes. They do. There is an enormous amount of variety in viruses and the types of cells they infect, so just as there are viruses that infect human cells, there are viruses called bacteriophages that prey on bacteria. Like other predators and their prey, bacteriophages and bacteria are locked in a bitter evolutionary arms race.
Virus vs. Bacteria

Before diving into the savage battle between bacteria and bacteriophages, let’s look at how it’s possible for a virus to infect a bacterium. Bacteria are quite self-contained. In addition to housing DNA, a bacterium has the machinery required for a fully functional metabolism so it can grow and divide to create more bacteria. In contrast, viruses at their simplest are genetic containers. Unable to replicate its own genome or make its own proteins, a virus must hijack a host cell (like a bacterium) in order to be fruitful and multiply. So while bacteria cause infection to find more resources to continue growing and dividing on their own, a virus turns infected cells into virus factories. Using the cell’s replication machinery, metabolism, and resources, the virus turns the cell into a mere zombie that churns out viral proteins, viral nucleic acids (DNA and RNA) and builds more viruses.
Meet the predator
Most bacteriophages, or phages for short, have a very distinctive structure built of protein that transports the virus’ genome between cells (Figure 1). Reminiscent of a rocket ship, a phage has three main components: a long, cylindrical tail attached to the head by a collar. The head, or capsid, is an icosahedron and built of many triangular segments that houses the virus’ nucleic acid (DNA). At the far end of the tail, a base plate, tail fibers, and tail spikes (not pictured) form a viral landing platform for the bacterial surface.
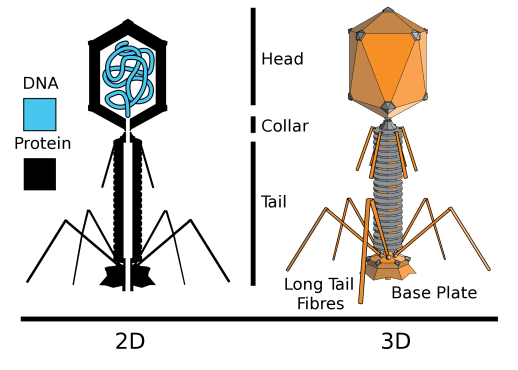
Devoured: How phages infect bacteria
The infection of a bacterium begins when the phage’s tail recognizes and binds to specific proteins at the surface of the cell, allowing attachment. Like a key fits a lock, the tail recognizes a specific bacterial feature (usually a protein) triggering the virus’s ability to inject its nucleic acid into the cell through the tail. Once in the cell, most viruses have two choices (Figure 2).
First, the viral DNA can zombie-fy the bacterium, tricking it into making more copies of the DNA and producing viral proteins. These proteins assemble around a copy of viral DNA inside the cell, producing a new virus. The bacterial cell continues producing viruses until it literally bursts (i.e. lyses), releasing them from the cell to infect new bacteria. In this lytic phase, the virus takes over and exploits the resources of the enemy garrison, then leaves it burning.
The second option is the lysogenic (or sleeper cell) phase. Here, the viral DNA integrates itself into the bacterial genome and lies in wait. If the bacterial cell becomes stressed and might die, due to either lack of nutrients or the presence of toxins, the viral DNA can leave the bacterium’s chromosome and begin the lytic phase. In the meantime, however, the viral DNA hitches a free ride into any new bacteria made when the original bacterium replicates. In effect, a single virus can use the lysogenic phase to infect many bacteria.
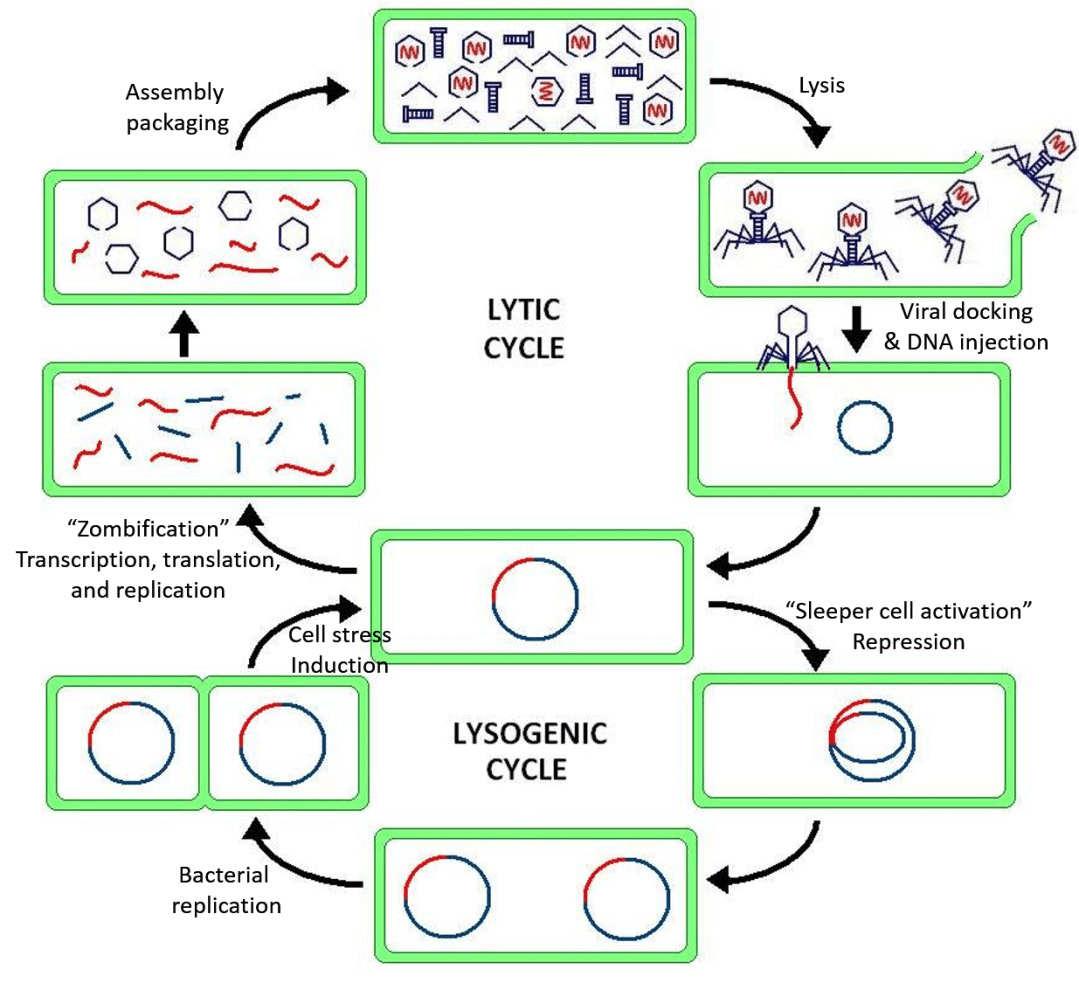
Escape strategies: Blocking bacteriophage infection
Like other prey, bacteria have evolved many ways to avoid predation. The most common method is by blocking viruses from docking on the bacterial surface, “changing the lock” so that the key no longer fits. Each time a bacterium replicates, a number of errors, or mutations, occur in its DNA. A mutation can be neutral, harmful, or occasionally beneficial—for example, changing the protein a phage binds to without damaging it, thus blocking phage infection of the bacterium. However, when the phage genome is replicated within a bacterium, the same mutation process might alter the viral DNA. This means a phage could acquire a mutation in its tail, allowing it fit the new lock. And so the predator adapts to the prey’s escape strategy.
Bacteria can also destroy the viral genome before it has a chance to hijack the bacterium’s machinery. Bacteria barcode their genome by adding patterns of methyl (1 carbon plus 3 hydrogens; CH3) tags to their own DNA. When bacteria encounter DNA with a different barcode, restriction enzymes chew up the foreign DNA. This restriction-modification system stops a number of phages from successfully infecting bacteria. In addition, some types of bacteria have the ability to “remember” and become immune to specific viruses using the CRISPR/Cas system. CRISPR is a section of the bacterial DNA where the bacterium stores short sequences of viral DNA, usually from when a phage was destroyed by restriction enzymes or when the cell encountered phage DNA from a cell that lysed nearby. Cas proteins bind copies of the saved DNA segments and use them to “survey” incoming DNA. If they match, then the incoming DNA is targeted by the cell to be degraded. But of course, there are bacteriophages with genes that block bacterial CRISPR/Cas systems.
The life or death battle between phages and bacteria has raged for millions of years, and will continue. However, it’s a war that we humans can use to our advantage. Next time, I’ll describe how researchers are using this mortal combat to help improve science and human health.
Read the next installment “Virus vs. Bacteria: Enemy of my enemy” here.
About the author
 Ada Hagan is a doctoral student here at the University of Michigan in the department of Microbiology and Immunology. She does recon on the sneaky ways bacteria find nutrients (like iron!) when they are invading our bodies. Originally hailing from the mountains of East Tennessee, Ada earned both her B.S. and M.S. in Microbiology from East Tennessee State University. In her spare time, Ada spends time with her pets and husband, cooking, fishing & the occasional Netflix binge. Follow her on Twitter (@adahagan) and see more posts from her on LinkedIn.
Ada Hagan is a doctoral student here at the University of Michigan in the department of Microbiology and Immunology. She does recon on the sneaky ways bacteria find nutrients (like iron!) when they are invading our bodies. Originally hailing from the mountains of East Tennessee, Ada earned both her B.S. and M.S. in Microbiology from East Tennessee State University. In her spare time, Ada spends time with her pets and husband, cooking, fishing & the occasional Netflix binge. Follow her on Twitter (@adahagan) and see more posts from her on LinkedIn.
Read more posts by Ada here.

7 thoughts on “Virus vs. Bacteria: Mortal combat”