Author: Carrie Johnson
Editors: Ada Hagan, Irene Park
Whether you have heard about it or not, antibiotic resistance is a growing threat that affects us all.
For generations, we have benefited from antibiotics to fight bacterial infections that would otherwise threaten our lives. Unfortunately, the effectiveness of antibiotics is increasingly at risk. Bacterial infections resistant to antibiotics already have already taken a significant toll and the severity of the problem is only growing. In the United States, it already costs us over 23,000 lives and an estimated $55 billion each year.
As we head into a new school year and the colder winter months when illness risks seem to rise, the timing couldn’t be better to remind you that everyone (yes, you!) plays a role in combating this growing problem of antibiotic resistance. But first we need to understand the basics of this problem, including the three major factors at play.
Economic constraints
Research is expensive. It costs nearly $2.6 billion to get a drug to market. With such high costs, pharmaceutical companies typically try to develop drugs that will provide a high return on investment. Consequently, companies seek to create drugs that patients take for long periods of time and never lose effectiveness, such as drugs for high cholesterol or diabetes, to maximize profit.
Antibiotics, on the other hand, are typically used for only one to two weeks. Furthermore, resistance spreads quickly, rendering the drug ineffective sooner and therefore prescribed less. This combination of short duration of consumption by a patient and the short lifetime a drug will work creates an economical double-edged sword. Thus, the incentives to develop new antibiotics are limited and the number of new antibiotic drugs approved by the FDA over the last two decades is worryingly small.
The good news is funding for antibiotic research is beginning to reflect our growing awareness of the problem. The newly approved budget for NIH includes an additional $303 million to combat antibiotic resistance and scientists are developing techniques and technology to discover new potential antibiotics.
Bacterial biology
There are many classes of antibiotics with diverse methods for killing bacteria, but bacteria can quickly acquire resistance. The very nature of antibiotics is that the more they are used, the more bacteria are exposed to them, and with more exposure comes more opportunities for resistant bacteria to thrive.

When bacteria are exposed to an appropriate antibiotic, nearly all of them die. Unfortunately, because bacteria are numerous and reproduce quickly, there is a high probability that one or more of them could already have a mutation in their genome that allows them to avoid death by the antibiotic. These resistant bacteria grow and divide rapidly while the others without the mutation die. All it takes is one resistant bacterium to thrive. If you haven’t seen it yet, you are highly encouraged to check out this incredible new video that shows antibiotic resistance developing right in front your eyes.
Compounding the problem of antibiotic resistance amongst one bacterium is the fact that bacteria can also share DNA that contains instructions on how to survive the antibiotic. This mobilization of DNA allows for resistance to spread, even between bacterial species. These DNA mutations have led to many different tools that allow bacteria to evade antibiotics. It also enhances the chances of creating “superbugs”—bacteria with resistance to many antibiotics.
Together, rapid growth, mutations, and the ability to share DNA are properties inherent to bacteria and cannot be avoided. As humans, what we can do is slow this process down by limiting the exposure of bacteria to antibiotics to give them fewer chances to develop resistance in the first place. This leads us to the last major factor to consider in antibiotic resistance.
Consumer misuse
If we are going to slow this process down, we need to use antibiotics appropriately. Unfortunately, in agriculture and in our own personal health behavior, we are doing a very poor job of doing this.
Due to the systematic and inappropriate administration of antibiotics, an estimated 70-80% of all antibiotics in the US are used in animals. Despite not being sick, entire flocks or herds are given consistent, low doses of antibiotics to promote their growth on less feed. The low doses are not enough to fully kill the bacteria, allowing for selection of resistant bacteria (Figure 2). The antibiotic use in agriculture is not well regulated, where their administration is primarily done in the name of speeding up the growth of animals to getting more of them to market sooner, but it is also done so at the cost of future human health. From the Center for Disease Control to the American Academy of Pediatrics to the Food and Drug Administration, many are recognizing this practice needs to stop and needs regulation so that only animals that are actually sick receive antibiotics to curb growing resistance. Countries such as Denmark have already banned antibiotic use for agricultural growth promotion, successfully reducing antibiotic consumption while actually seeing an improvement in agricultural productivity.
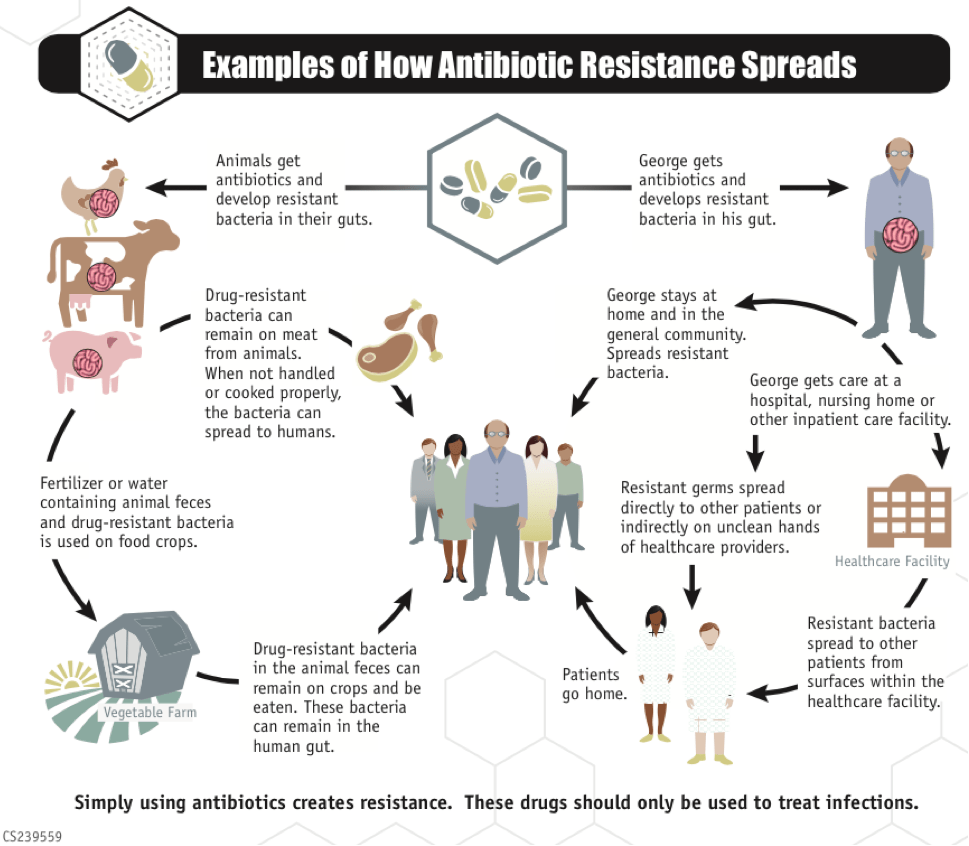
In humans, antibiotic misuse happens in a variety of ways. The most common way is when patients are given antibiotics when they are not needed. It is estimated that nearly 50% of all prescribed antibiotics are unnecessary. This preventable prescribing often happens when patients demand antibiotics from their doctor despite the infection being viral and thus untreatable by antibiotics. It is often just coincidence that such a patient starts to feel better on antibiotics as the virus runs its course and the immune system is able to overcome it. Antibiotics will not treat the cause of the infection and will simply expose the patient’s bacterial flora to the antibiotic, needlessly selecting for resistant bacteria (Figure 2).
Another area of misuse occurs when doctors prescribe an inappropriate antibiotic. Some antibiotics target a broad spectrum of bacterial species, including those that are not causing the infection, selecting for resistance in these other bacteria as well. These broad-spectrum antibiotics also kill some of the good bacteria in your digestive system and actually cause you more harm.
Finally, even when antibiotics are properly prescribed, some patients do not finish the prescribed course of antibiotics after they begin to feel better. This allows some bacteria to survive the sub-lethal dose, selecting for resistance (Figure 2).
Combined, the agricultural and human antibiotic misuse is a major contributor to this surging threat. However, this is the area we have the most control over and where we could have the biggest impact on subduing the problem of antibiotic resistance.
What can be done?
It can’t be stressed enough: the increasing resistance of bacteria to existing antibiotics combined with the lack of new classes of antibiotics presents a very real problem for our health. Infections caused by bacteria resistant to antibiotics are not only an inherent danger to patients who obtain infections from cuts and scrapes in everyday life, but they also threaten the way we treat other health issues. From cancer treatments to routine operations like C-sections and knee surgery, antibiotic resistance threatens many other areas of modern health care. We all have a personal stake in this.
So what can you do?
- Only use antibiotics when you have a bacterial infection and be respectful of your physician’s attempts to ensure that it is indeed bacterial and not viral.
- Complete the entire course of any antibiotic you are prescribed.
- Support policies that look to curb antibiotic use in the agricultural sector, such as this and this.
- As a consumer, buy meat that comes from animals raised without sub-clinical doses of antibiotics.
- Support policies and funding measures that promote research in new antibiotics, especially those looking to develop new classes of antibiotics.
- Take what you’ve learned from this article and educate others about this issue. Inform them about the need for responsible antibiotic use and further research to ensure the future of medicine is not taken for granted.
Antibiotic resistance is inevitable, but if we take measures to ensure proper antibiotic used and we invest in new ones, we can extend the lifetime of antibiotic drugs and limit the threat of untreatable, life-threatening bacterial infections.
About the author
 Carrie Johnson is a PhD student in Chemical Biology at the University of Michigan. Her research is on advanced prostate cancer and focuses on studying the role and regulation of specific proteins that help the cancer cells avoid existing therapies. Carrie’s undergraduate degrees in chemistry and biochemistry are from Gustavus Adolphus College in Minnesota, where she graduated in 2012. When not doing research in the lab, Carrie enjoys being active and playing many sports, being highly involved in STEM outreach through the FEMMES organization on campus, cooking and baking, and playing new board games with good friends and food. You can connect with her on LinkedIn here: www.linkedin.com/in/carriemjohnson. Read all posts by Carrie here.
Carrie Johnson is a PhD student in Chemical Biology at the University of Michigan. Her research is on advanced prostate cancer and focuses on studying the role and regulation of specific proteins that help the cancer cells avoid existing therapies. Carrie’s undergraduate degrees in chemistry and biochemistry are from Gustavus Adolphus College in Minnesota, where she graduated in 2012. When not doing research in the lab, Carrie enjoys being active and playing many sports, being highly involved in STEM outreach through the FEMMES organization on campus, cooking and baking, and playing new board games with good friends and food. You can connect with her on LinkedIn here: www.linkedin.com/in/carriemjohnson. Read all posts by Carrie here.
Image Credit:
Figure 1: CDC, Image source.
Figure 2: CDC, Image source.

Informative and interesting article Carrie, nice job.
LikeLike
Great article! I’m gonna never miss another article from misciwriters. You guys are providing such a valuable content for all the people on the internet to gain the knowledge regarding health issues and topics such as this article (antiobiotics resistance). Great work.
LikeLike