Author: John Charpentier
Editors; Noah Steinfeld, Tricia Garay, and Scott Barolo
A glance into any organic chemistry or biochemistry textbook reveals a dizzying variety of chemical compounds, reactions and mechanisms. It is not at all obvious why one particular class of reaction, the attachment and detachment of a phosphate group (PO43-) to molecules like nucleotides and proteins, is central to making the chemistry of life “go.”
Proteins: Not Just for Getting Swole, Brah

Proteins are the working-class heroes of the cell: they get things done. A protein’s function is largely determined by its shape, which in turn is dictated by the linear sequence of chemically distinct amino acid subunits it is composed of. The rules of protein folding are astonishingly complex. Generally speaking, the reluctance of hydrophobic (“water-fearing”) amino acids to project outward into the watery cytoplasm is the primary determinant of protein shape, but electrostatic interactions between amino acid residues are also important. Phosphate groups have three negative charges, which means that when they are linked to or removed from a protein by specialized enzymes, they can dramatically modify its shape and stability, and therefore its function. The phosphorylation/dephosphorylation cycle operates like a switch to regulate protein behavior: add a phosphate and you get a violent Mr. Hyde protein; take it off and you get the amiable Dr. Jekyll.

So where do we find phosphorylation in biochemistry? The answer is: pretty much everywhere! I will discuss two key examples. Firstly, phosphorylation is important in “cell signaling,” the sensing of messages from outside a cell and their incorporation into cellular decision-making. It’s worth observing that there isn’t anything we’d recognize as a brain in cells – decision-making is an emergent property of the integration of these signals, not the doing of a microscopic cellular homunculus pulling levers or “thinking.”
Placing a “Cellular” Phone Call
To understand the role of phosphorylation in cell signaling, we’ll consider a specific cell signaling pathway called the receptor tyrosine kinase (RTK) pathway. RTK signaling typically begins when a small molecule binds to a receptor protein on the outer membrane of a cell. This causes a change in the shape of the receptor, revealing other protein surfaces that can then be phosphorylated by kinases that hang out nearby. When the tail of the receptor, which sticks out of the inner surface of the membrane, is phosphorylated, an adaptor protein is recruited to it, and the two proteins click together like Lego pieces. This sets off a cascade of phosphorylation, in which kinases are phosphorylated by other kinases, causing them to become active and phosphorylate still more kinases (see Figure 3).
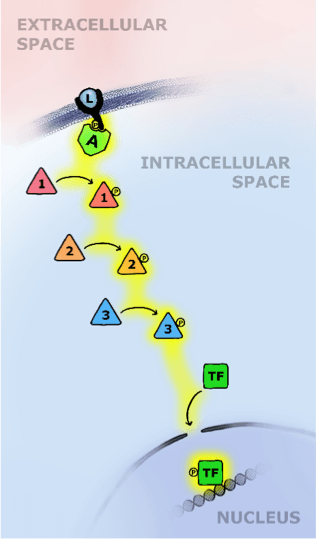
The cascade ends when a protein called a transcription factor is phosphorylated, allowing it to move into the cell’s nucleus, bind to DNA, and turn genes on or off. Cell signaling occurs on the order of minutes to hours, so there are plenty of opportunities for other proteins to redirect, modify or block the message at any point, like hanging up on a call.
Paying the Bills
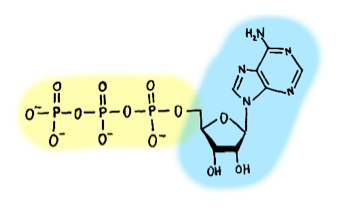
Phosphates are also crucial components of the energy currencies in cells, adenosine triphosphate (ATP) and guanosine triphosphate (GTP). These molecules resemble the single units (nucleotides) that make up DNA and RNA, and may have evolved from them. They consist of a small, cyclic sugar attached to a “nucleobase” (adenine or guanine) to form a nucleoside (adenosine or guanosine). To these are added a chain of three linked phosphate groups. Water molecules can be added to break the high-energy bonds between each phosphate group, releasing energy that nearby enzymes can use to do work. Without ATP and GTP, synthesis of the molecules that make up living organisms, and their organization into cells, would be impossible! In fact, the reason why we need to eat to live is so that we can steal high-energy electrons from our food and use them to regenerate the ATP dollars.
But that’s a subject for another post…
Why Phosphates?
So now we arrive at the question we opened with: why phosphates? Though other chemical groups are reversibly attached to proteins to regulate their behavior, stability, degradation, and trafficking within the cell, none are as ubiquitous or arguably as important as phosphorylation. Apart from storing large amounts of energy in the bonds linking them, do phosphate groups have chemical properties that make them especially desirable modifications?
Phosphate groups have three negative charges, which make it nearly impossible for them, or the proteins to which they are attached, to diffuse through cellular membranes and be lost from the cell. Phosphate linkages are also extremely stable – the half-life of a single ester phosphate linkage is estimated to be about 1012 years!
Phosphates have been important for quite a while in our evolutionary history. For a bond to occur between two DNA or RNA subunits, a pair of electrons must be donated to each subunit—these come from a phosphate. After linking the adjacent nucleotides, two negative charges will be neutralized, so an additional negative charge is needed to prevent diffusion through the membrane (see Figure 5). Phosphate groups have the required three, allowing DNA and RNA molecules to stay in the cell.

It’s possible phosphates were first used to link DNA and RNA nucleotides long before the first living cell reproduced itself. Perhaps during the prebiotic chemical cycles that took place between 4.5 billion years ago, when Earth was formed, and the appearance of the first cells about one billion years later. How phosphorylation came about, and what its first job was, remain the subjects of vigorous scientific debate.
One thing is for certain, though: humble phosphates and all living things have been inextricably linked ever since, relaying messages, paying entropic debts, and enabling microscopic industries of all kinds.
About the author
 John Charpentier is a first-year Immunology Ph.D student at the University of Michigan. Prior to beginning graduate school, John earned a B.S. in biochemistry at the University of Massachusetts Boston. John is investigating a form of endocytosis (the transport of fluids and molecules) that occurs in activated T cells. Outside of the lab, he enjoys visiting museums, attending plays and concerts, and is developing a podcast about great experiments in the history of biological research. Follow him on Twitter (@snpsandsnRNPs), and connect on LinkedIn.
John Charpentier is a first-year Immunology Ph.D student at the University of Michigan. Prior to beginning graduate school, John earned a B.S. in biochemistry at the University of Massachusetts Boston. John is investigating a form of endocytosis (the transport of fluids and molecules) that occurs in activated T cells. Outside of the lab, he enjoys visiting museums, attending plays and concerts, and is developing a podcast about great experiments in the history of biological research. Follow him on Twitter (@snpsandsnRNPs), and connect on LinkedIn.
Read all posts by John here.
Image credits: All figures were generated by the author.
