Author: Jessica McAnulty
Editors: Tricia Garay, Stephanie Hamilton, and Whit Froehlich
Most likely, you know of someone diagnosed with breast cancer, which affects 1 in 8 women in the United States. Some of the reasons this disease is so difficult to treat are the lack of targeted therapies (as there are different subtypes of breast cancer) and tumor resistance to treatment. Therefore, scientists are investigating novel therapies that act on a specific component of the cancer and/or prevent this resistance. One exciting therapy alters the expression of certain genes; a gene needs to be expressed, or “turned on”, in order for the cell to obtain information from the gene and produce a product. This therapy is a promising approach since cancers, such as hormone-sensitive breast cancer, are often due to genetic mutations that result in an increase in gene expression. It is thought that using this therapy to alter gene expression will reverse the breast tumor’s resistance to treatment.
Hormone-sensitive breast cancer subtypes
Identifying a breast cancer patient’s cancer subtype is necessary in order to provide a targeted treatment. Some breast cancers are sensitive to estrogen and progesterone, natural hormones found within the body, due to an overexpression of hormone receptors on the tumor. Upon diagnosis, a doctor will test which receptors a patient’s tumor has. The two most common receptors are estrogen receptor (ER) and progesterone receptor (PR). 80% of breast cancer cases are ER positive, meaning the ER gene is expressed and therefore the tumor cells have ERs. Within the ER-positive group, a majority of cases are also PR positive. Because ER and PR commonly occur together, this combination is denoted as hormone receptor (HR) positive.
Expression of receptors on the tumor cells encourages tumor growth. Therefore, understanding which receptors are expressed is important in determining the proper treatment for the patient. Some targeted hormone therapies have been successful for HR positive breast cancer by inhibiting the body’s production of the hormones. However, tumors can develop resistance to these therapies, and the cancer remains. Scientists suspect that modifications of gene expression may have a role in therapy resistance. Therefore, several scientists are working to develop a drug that manipulates gene expression.
DNA Organization & Epigenetics
In order to understand how gene expression can be manipulated, it is important to know how DNA is stored. Each cell in the body contains a human’s full genetic code, which is made of about 3 billion base pairs and reaches about 6 feet in length if it’s completely stretched out. If the DNA from every cell in the body was connected, it would reach 328 trillion feet, enough to circle the Earth 2500 times! So how does all this DNA fit in the body? DNA goes through a multi-stage folding process; the first crucial step is the wrapping of DNA around proteins called histones. When DNA wraps tightly around the histones, the cell’s machinery cannot access the gene, and it cannot be used. On the other hand, if the DNA is exposed from the histone, the cell’s machinery can read the gene and it can be expressed. The fate of the gene’s expression is determined by certain chemical tags on the DNA or histones that alter how tightly the DNA wraps around the histones. These chemical tags that affect gene expression are considered to be an epigenetic modification.
“Epi” is a Greek prefix meaning “on” or “above”. Therefore, epigenetics is the study of modifications on the gene, distinct from direct changes to the genetic code — the addition of an epigenetic tag never alters the DNA content itself. Importantly, these tags are not permanent and can be added or removed by various enzymes. One common chemical tag to turn a gene on is histone acetylation (Figure 1, right image). This is the process of adding an acetyl group — a combination of carbon, hydrogen, and oxygen molecules — to the histone. An enzyme called histone deacetylase (HDAC) can reverse the acetylation, removing the chemical tag and turning the gene off (Figure 1, left image).
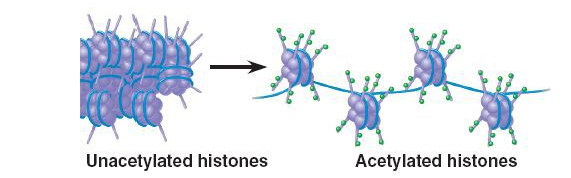
Figure 1. The blue strand represents DNA; histones are the clusters of purple spheres. DNA wraps tightly around histones when the histones are unacetylated. In this case, the gene is off and inaccessible. However, when the chemical tag (represented by the small green spheres) is added to the histones, the DNA unwraps from the histone and the gene is turned on. If an HDAC was added, it would remove the acetylation tag and the gene would return to the unacetylated state (left).
Epigenetic therapeutics for breast cancer
Scientists are now investigating the effects of inhibiting HDAC on breast cancer. If a drug blocks the HDAC, the histone remains acetylated and the gene will stay on. This manipulation of the DNA and histone structure by the HDAC inhibitor can affect expression of genes that repress cancer, resulting in cancer cell death. The hope is that these HDAC inhibitors will take advantage of the tumors’ hormone sensitivity, reversing treatment resistance and improving efficacy for more patients. There have been several clinical trials that investigate the HDAC inhibitor on its own, or in combination with another therapeutic. For example, Vorinostat, an FDA approved HDAC inhibitor that treats lymphatic cancer, is used in a current clinical trial to investigate the effects of Vorinostat combined with hormone therapy on breast cancer patients. Entinostat is another HDAC inhibitor that is being evaluated as a therapy for several cancers, including ER positive breast cancer. An ongoing phase three trial pairs Entinostat with hormone therapy for HR positive patients. If these trials show that pairing a HDAC with another therapeutic is successful in making the tumor more sensitive to treatment, then there is a possibility that this approach might apply to other cancers and other therapeutics.
These HDAC inhibitors just scratch the surface of anti-cancer epigenetic therapeutics. Histone acetylation is one of several epigenetic tags, and HDAC is one of many enzymes with epigenetic functions. Some of these other enzymes have also been identified as anti-cancer therapeutics! For example, a different epigenetic enzyme received FDA approval and is used to fight against blood cancer. There are even clinical trials that pair two epigenetic enzymes and measure their effects on lung cancer. Therefore, as more is understood about epigenetics, it might be possible to discover new epigenetic therapeutic targets. The range of epigenetic therapeutics and various clinical trials already shows a promising start. Hopefully, this growing field of epigenetic therapeutics will ultimately reduce the impact of cancer.
 Jessica earned her BS in Biological Sciences from the University of Delaware. During her undergraduate, she conducted research on behavioral epigenetics and completed two pharmaceutical internships. Through these experiences, she became interested in epigenetics and disease, and she is now pursuing her PhD in pathology at the University of Michigan. Aside from learning, Jessica enjoys adding houseplants to her growing collection, exploring Michigan’s beautiful scenery, and driving her Mini Cooper.
Jessica earned her BS in Biological Sciences from the University of Delaware. During her undergraduate, she conducted research on behavioral epigenetics and completed two pharmaceutical internships. Through these experiences, she became interested in epigenetics and disease, and she is now pursuing her PhD in pathology at the University of Michigan. Aside from learning, Jessica enjoys adding houseplants to her growing collection, exploring Michigan’s beautiful scenery, and driving her Mini Cooper.
